Coronaviruses include some pathogens that are fatal to humans and animals, such as severe acute respiratory syndromes (SARS) and Middle East respiratory syndrome (MERS), which cause devastating diseases and tremendous loss. Infectious bronchitis virus (IBV), the avian coronavirus, is economically important to the poultry industry worldwide, and the continuous emergence of new viral variants complicates its disease control. There is an urgent need to characterize the emerging variants and to develop more effective countermeasures for disease management.
Two important studies have been completed by a team at the Department of Veterinary Medicine, National Taiwan University, led by Assistant Professor Hui-Wen Chen. It was determined that since 2002, IBV variants have emerged through frequent recombination events among the Taiwan strains, China strains, Japan strains and vaccine strains, which has caused severe outbreaks in the field. After further investigation, one of the new variants, IBV 3575/08, was determined to carry mutations in non-structural accessory proteins such as 3a, 3b and 5b. Despite a high degree of homology in their spike protein genes, cross-neutralization tests have clearly suggested that IBV 3575/08 presents distinct antigenicity and cannot be protected against using conventional vaccines. Animal challenge experiments have exhibited strong and complex respiratory and renal pathogenicity for 3575/08. In addition, early and prolonged viral shedding and rapid viral dissemination have also been observed. Furthermore, IBV 3575/08 delayed the expression of a subset of early innate immune genes in chickens. The present study reveals a new IBV strain, which harbors a known local genotype but displays remarkably altered antigenicity, pathogenicity and host defenses.
On the other hand, the ongoing battle against current and rising viral infectious threats has prompted increasing efforts in the development of vaccine technology. A major thrust in vaccine research focuses on developing formulations with virus-like features towards enhancing antigen presentation and immune processing. We have developed an easy approach to formulating synthetic virus-like particles (sVLPs) and exploited the phenomenon of protein corona formation induced by the high-energy surfaces of synthetic nanoparticles. Using an IBV spike protein as the model antigen, sVLPs were prepared by incubating 100 nm gold nanoparticles in a solution containing an optimized concentration of viral proteins. Compared to inoculation with free proteins, vaccination with the sVLPs showed enhanced lymphatic antigen delivery, stronger antibody titers, increased splenic T-cell response, and reduced infection-associated symptoms during infection. A comparison to a commercial whole inactivated virus vaccine also showed evidence of superior antiviral protection by the sVLPs. This study demonstrates a simple yet robust method for bridging viral antigens with synthetic nanoparticles for improved vaccine application. Given the robustness and versatility of the approach, it can be envisioned that the technique can be broadly applied for the development of different vaccines.
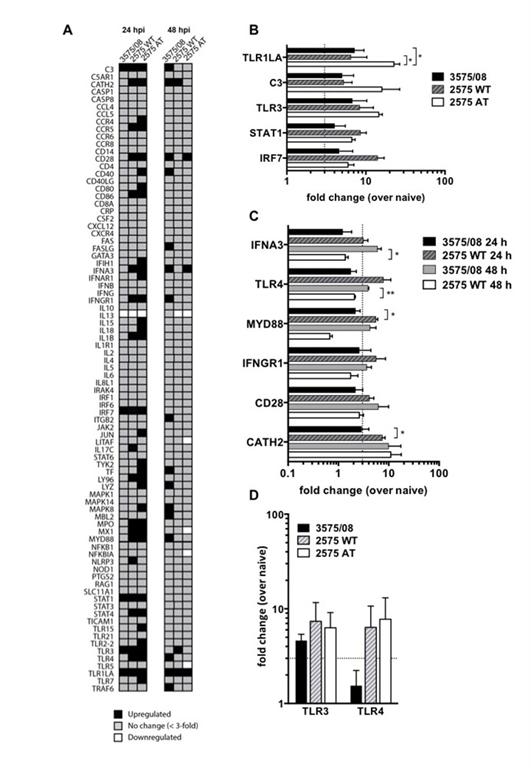
Figure 1. Differential innate immune responses induced by various IBVs (left).
Figure 2. Illustration of the synthetic virus-like particles for anti-IBV vaccination (right).
References
1. Hui-Wen Chen, Chen-Yu Huang, Shu-Yi Lin, Zih-Syun Fang, Chen-Hsuan Hsu, Jung-Chen Lin, Yuan-I Chen, Bing-Yu Yao and Che-Ming J. Hu. (2016). Synthetic Virus-like Particles Prepared via Protein Corona Formation Enable Effective Vaccination in an Avian Model of Coronavirus Infection. Biomaterials. 106, 111-118. DOI:10.1016/j.biomaterials.2016.08.018.
2. Shu-Yi Lin, Yao-Tsun Li, You-Ting Chen, Ting-Chih Chen, Che-Ming J. Hu, and Hui-Wen Chen. (2016). Identification of an infectious bronchitis coronavirus strain exhibiting a classical genotype but altered antigenicity, pathogenicity, and innate immunity profile. Scientific Reports. 6:37725. Published online on November 23, 2016. DOI:10.1038/srep37725.
Assistant Professor Hui-Wen Chen
Department of Veterinary Medicine
winnichen@ntu.edu.tw